Mechanistic studies on the human mitochondrial chaperonin mtHsp60 and its pathological mutations |
||
|---|---|---|
|
Our current research focuses on investigating the molecular mechanisms by which the mitochondrial chaperonin mtHsp60 maintains mitochondrial protein homeostasis and contributes to human diseases. mtHsp60 has been associated with various health conditions, including Alzheimer�s disease and cancer. Mutations in mtHsp60 are responsible for some cases of neurodegenerative diseases. Because of its central role in disease processes, mtHsp60 has been proposed as a biomarker for diagnosis/prognosis and a drug target. However, our understanding of the mechanism of mtHsp60 is limited, mainly due to the challenges associated with its purification.
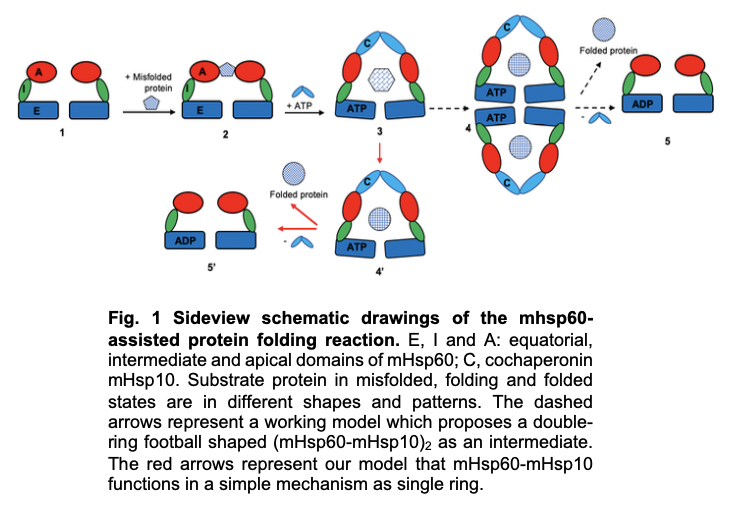
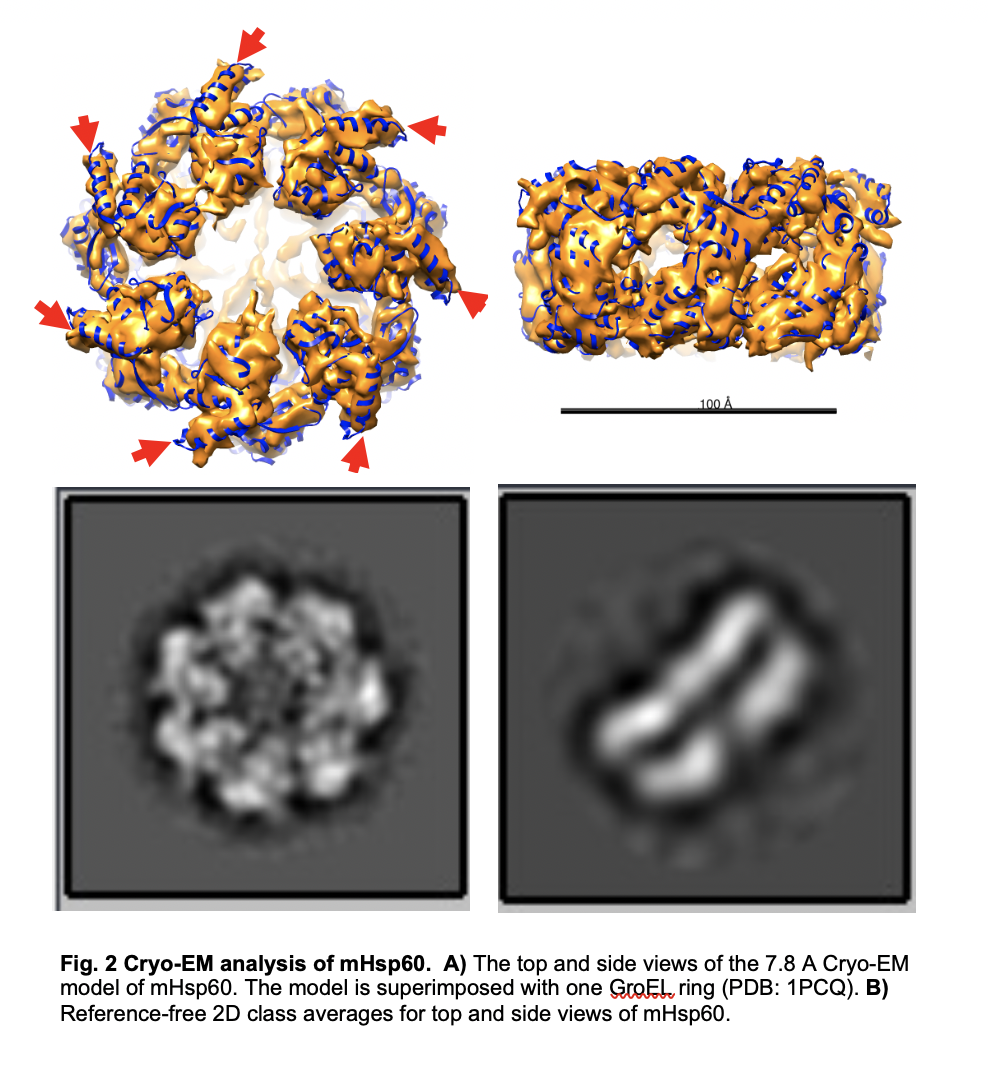
We have developed a protocol to purify active mHsp60 directly, without the need for refolding and reconstitution steps. We are using a range of biochemical, in vivo, and structural techniques, particularly cryogenic electron microscopy (cryo-EM), to elucidate the molecular mechanisms of mtHsp60 and its pathological mutations. One of our ultimate goals is to identify molecular targets for therapeutic development. So far, we have determined the cryo-EM structure of mtHsp60, providing a structural basis for its unique properties. Unexpectedly, we found that a pathological mutation increases both the structural stability and biochemical activity of mtHsp60. We are actively investigating the molecular steps impacted by the mutation and how these effects can be rectified by small molecules. Our studies will contribute to the mechanistic understanding of mtHsp60 and its associated disease processes, as well as the development of mtHsp60-based therapeutics.
|
Copyright � Chen Research Group, Department of Molecular and Cellular Biochemistry, Indiana University
Maintained by Adhitya Balaji (last updated May 23, 2020)
Back to previous page